Stop slow budget negotiations from stalling your trial – activate sites quicker, enroll subjects sooner, and stay ahead of timelines.
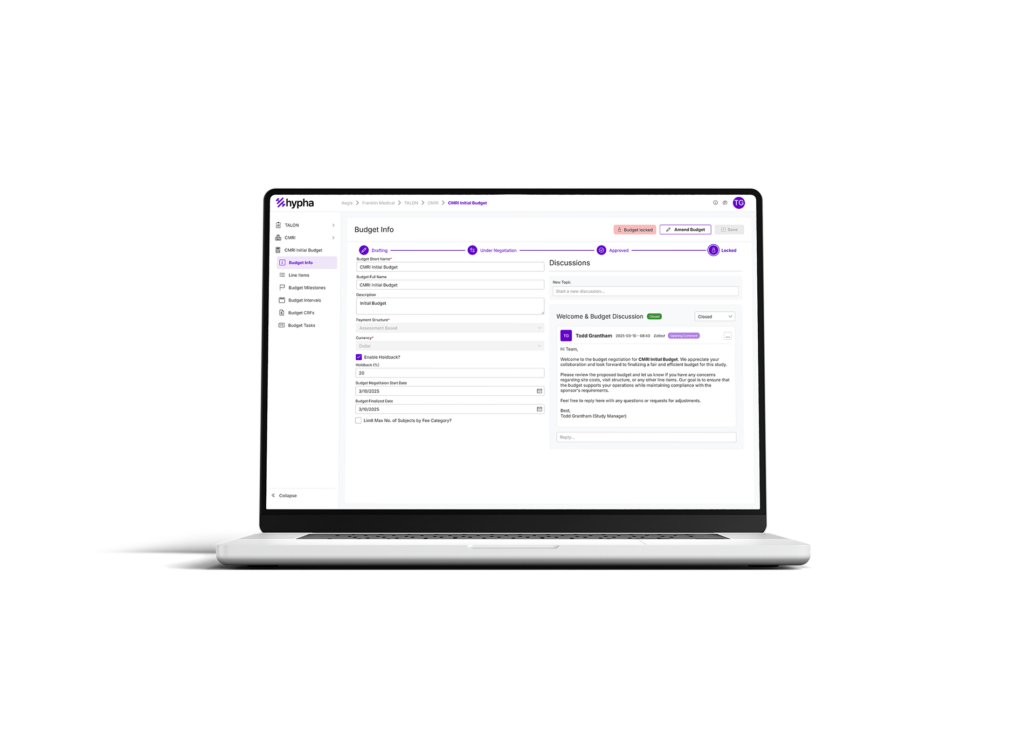
Every day that budget approvals are delayed, site activation stalls—slowing down enrollment and pushing trial timelines further out. Your team is stuck in manual processes—endless emails, Excel spreadsheets, and back-and-forth approvals—while sponsors and internal teams demand faster results.
The cost? Delayed negotiations, frustrated sites deprioritizing your study, and longer timelines that put your trial at risk. But it doesn’t have to be this way. A faster, more efficient and affordable process is within reach with Hypha!
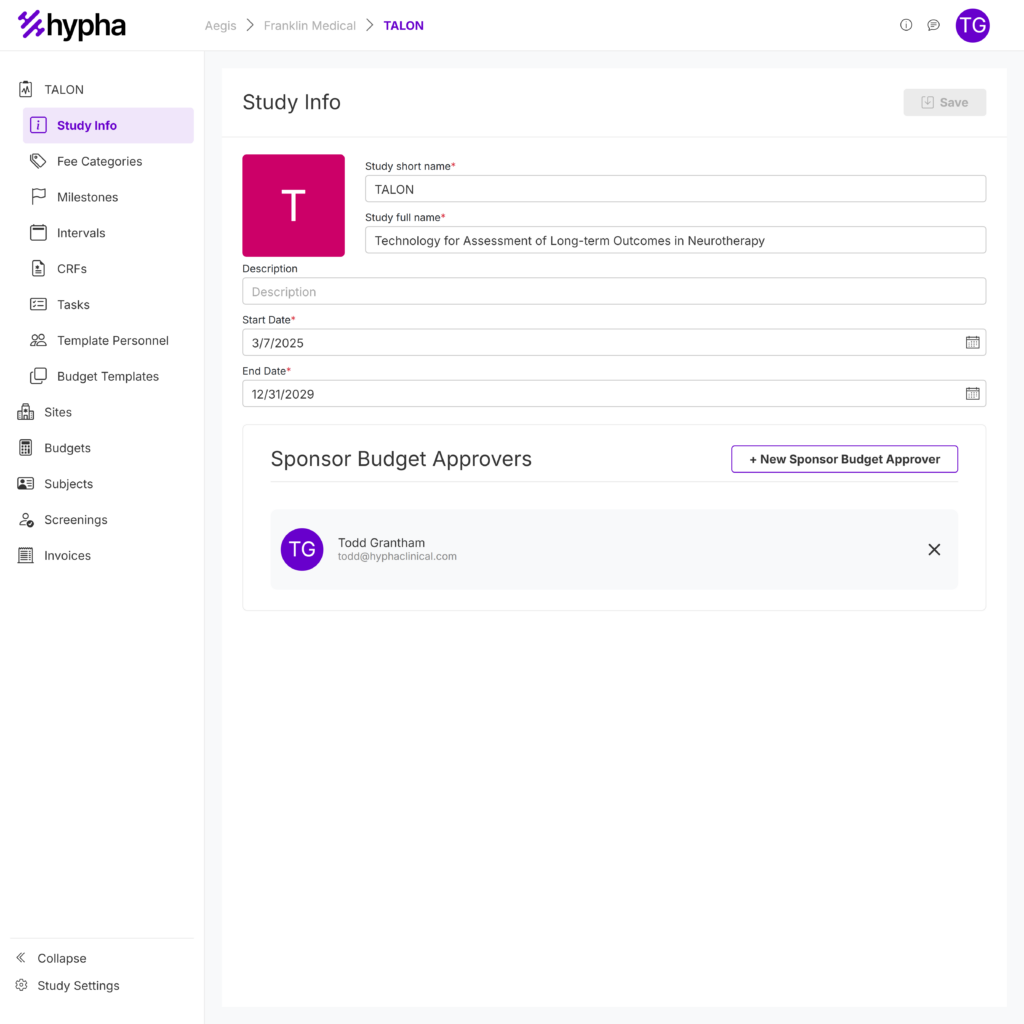


Is your team stuck managing approvals through outdated systems that weren’t built for clinical trial budget management? With years of experience in clinical trial budgeting and invoice processing, we created the Budget Manager to eliminate these roadblocks. Hypha streamlines budget review and approvals, and ensures faster contract execution—so your trials stay on schedule.
The more efficient way to negotiate and manage trial budgets is here. Get your trial moving with Hypha.
Real-time collaboration and communication tools so you can finalize budgets without endless email chains.
Unified communication threads (by budget or fee-specific) to keep teams, sites, and sponsors aligned, which means less confusion and smoother negotiations. Manage multiple site negotiations simultaneously.
We all know budgets may need to be amended from time to time. Adjust site budgets instantly, while always maintaining an accurate budget and invoicing history.
Submit your business email, and we’ll review your request—most approvals happen within 24 hours, so you can start exploring the Budget Manager in no time. (One complimentary study per CRO/Sponsor.)

Create your budget templates and clone for individual, customizable site budgets. Your first study is free!

Eliminate back-and-forth delays, improve financial accuracy, and speed up approvals—all on your own terms.

Track, update, and approve budgets in one place—no more version confusion, scattered spreadsheets or months of email chains.
Streamline the negotiation and approval process to reduce delays and speed up site activation.
Compare multiple budgets at a glance to make data-driven decisions faster.
Give all stakeholders secure, role-based access to budgets, ensuring transparency and alignment.
Customize budgets per site to account for regional cost differences and unique agreements.
Control when payments are released based on site progress, ensuring compliance and financial accuracy.
Once your request is approved, you can start setting up your budget templates within minutes. The approval process is manual, but once granted, you’ll have full access to start working immediately. While there’s a short learning curve, our platform is built specifically for CROs and sponsors, making it intuitive and easy to adopt.
No! The Budget Manager is web-based and requires no IT involvement or installations. It’s designed for clinical trial teams, not tech experts.
Budget Manager is completely free for CROs and sponsors for your first trial. Sites also get free access for trial budgets built and managed in Hypha. There’s no financial risk—just request access and start building your budgets today.
We built Hypha specifically for small to mid-sized CROs and sponsors, ensuring it meets the exact challenges you face in budgeting, approvals, and payments.
Yes! Sites get free, direct access, allowing them to review budget proposals, collaborate in real time, and negotiate faster. This reduces back-and-forth emails and delays while keeping everything documented in one place.
No, it’s the opposite! Budget Negotiator automates tracking, organizes negotiations, and eliminates redundant emails, reducing workload while improving efficiency.
Yes! Customizable budget templates ensure consistency, speed, and fewer manual errors.
Yes! Granular budget approvals give you full control over financial alignment before moving forward.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.